Rho Puts the Brakes on RAS-Driven Liver Cancer
MBI SCI COMMS | JulY 2014
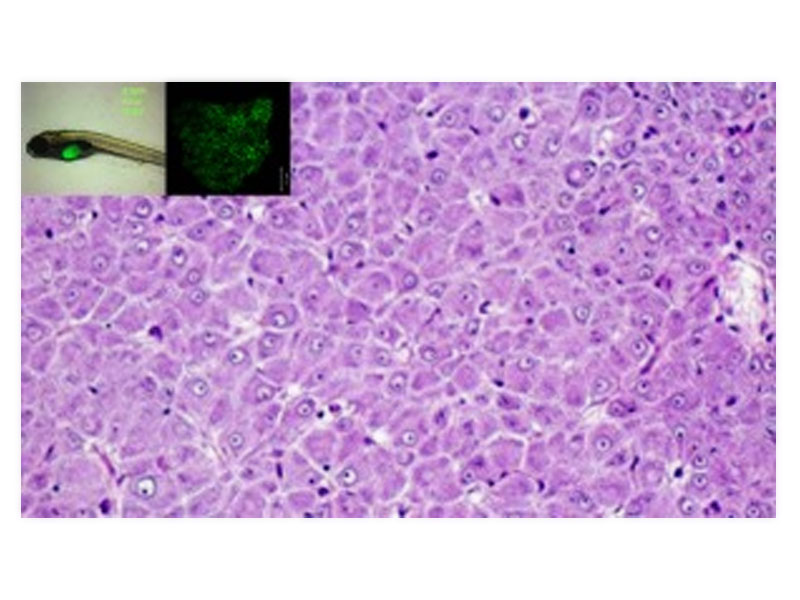
Assoc. Prof. Low Boon Chuan, principal investigator at MBI, and his team have identified a cross-talk between Ras and another signaling protein, RhoA, in zebrafish liver cancer models. Rho regulates signaling pathways critical for actin reorganization. As RhoA levels and activity are often elevated in various tumour types, including liver cancer, RhoA is thought to play a crucial role in cancer progression.
Chew TW, Liu XJ, Liu L, Spitsbergen JM, Gong Z, Low BC. Crosstalk of Ras and Rho: activation of RhoA abates Kras-induced liver tumorigenesis in transgenic zebrafish models. Oncogene. 2014, 33(21):2717-27.
Clues for targeting Ras-driven liver cancer through RhoA activation
The human body is made up of microscopic units called cells. Individual cells assemble into tissues and organs during the earliest stages of our development.
Normally, cells divide in a controlled manner to support growth, and when cells become old or damaged, they are removed through a process of programmed cell death. Cell division as well as cell death is regulated by multiple mechanisms, and perturbations in these mechanisms may cause cells to divide uncontrollably and evade cell death, resulting in cancer.
Cell signaling mechanisms allow cells to convert chemical or mechanical signals from various external stimuli into specific cellular functions, including cell growth and proliferation. Cell signaling is complex and consists of multiple pathways that involve a number of proteins. These ‘molecular switches’ play critical roles by relaying signals at the membrane through the cytoplasm into the cell’s nucleus where the genetic material is read and decoded. Examples of molecular switches include proteins belonging to the Ras family of small GTPases like Ras and RhoA proteins.
Ras and Rho in cancer progression
The Ras protein mediates cell signaling to regulate cell growth and survival. This is achieved primarily via the MAPK and the PI3K/Akt pathways. Mutations in Ras, and aberrant Ras signaling pathways, are frequently associated with cancer. Therefore Ras is an obvious target for anti-cancer drug discovery.
Yet, despite numerous efforts to target Ras, it remains an ‘undruggable’ protein, and attempts to target down-stream players of Ras signaling have not yielded therapeutic success. Although Ras is a protein that has been extensively studied, there are considerable knowledge gaps in understanding how Ras interacts with other proteins in vivo. Of particular interest is the cross-talk between Ras-mediated signaling, and other signaling pathways. Such knowledge gaps may conceal redundant pathways, feedback loops and other factors that enable Ras oncogenesis. Filling these gaps is crucial in the development of Ras targeted therapies.
Assoc. Prof. Low Boon Chuan, principal investigator at MBI, and his team have identified a cross-talk between Ras and another signaling protein, RhoA, in zebrafish liver cancer models. Rho regulates signaling pathways critical for actin reorganization. As RhoA levels and activity are often elevated in various tumour types, including liver cancer, RhoA is thought to play a crucial role in cancer progression.
Ras mutations, especially mutations in the Ras isoform K-Ras are implicated in the progression of hepatocellular carcinoma (HCC), which is a leading cause of death worldwide. Chew et al expressed a mutant form of K-Ras in zebrafish and observed the formation of liver tumors that closely resembled human HCC. This also coincided with the activation of Ras-mediated signaling pathways. However, when they simultaneously expressed an activated form of RhoA along with mutated K-Ras, tumor growth slowed down and survival increased. Conversely, depletion of RhoA augmented K-Ras driven tumor formation. Evidently, RhoA acts as a molecular brake on liver cancers driven and accelerated by Ras. This is an important discovery that may channel Ras-targeted cancer drug discovery efforts through RhoA.
Chew TW, Liu XJ, Liu L, Spitsbergen JM, Gong Z, Low BC (2014) Crosstalk of Ras and Rho: activation of RhoA abates Kras-induced liver tumorigenesis in transgenic zebrafish models. Oncogene. 33(21):2717-27 doi:10.1038/onc.2013.240
In the news
This research has been featured in the following news outlet:
- The Straits Times: Clue to slowing liver cancer lies in zebrafish