LOW Boon Chuan
Associate Professor, Mechanobiology Institute, National University of Singapore
dbslowbc@nus.edu.sg
Level 10 T-Lab
National University of Singapore
5A Engineering Drive 1
Singapore 117411
Research Program
The Cell-Matrix and Cell-Cell Mechanotransduction Group
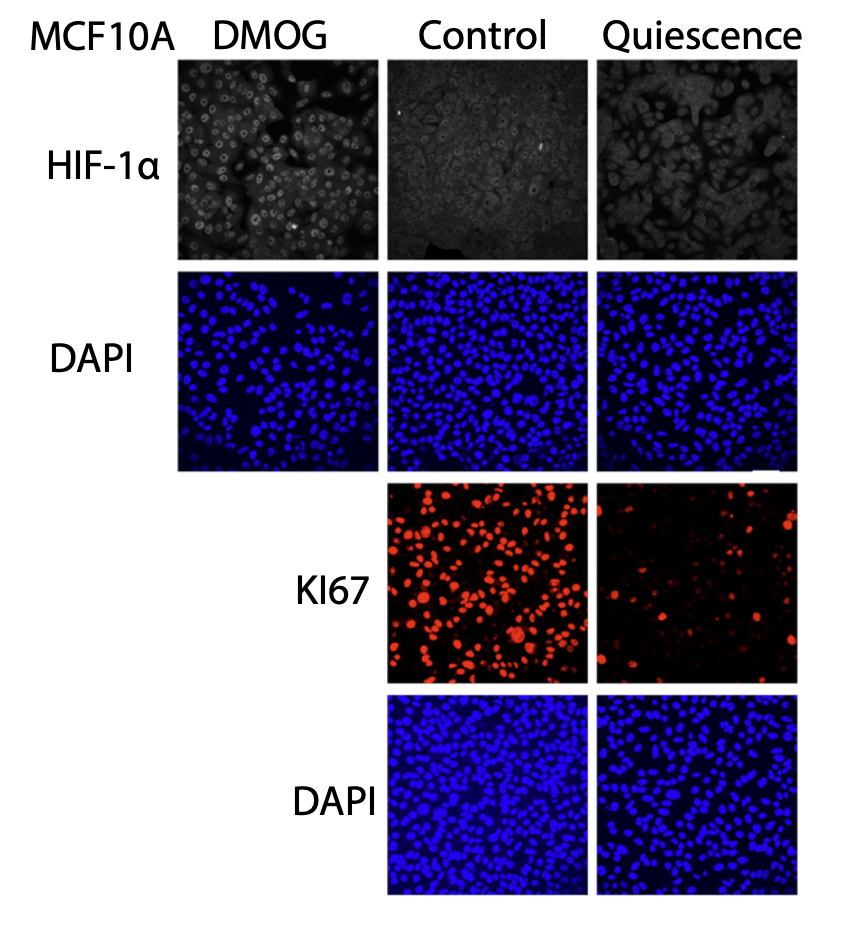
How Structural Imbalance Drives Inflammatory Signaling in Senescent Cells
In a study published in Molecular Biology of the Cell led by Celestine Ho at the Mechanobiology Institute, NUS, researchers discover that HIF-1α-activation in SASP is a defining feature of the SASP induced by diverse stressors, acting independently of micronuclei generation and cGAS/STING activation.
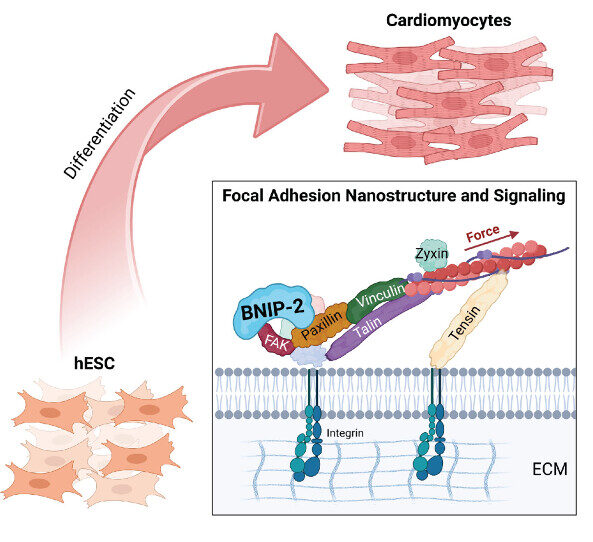
The Nano-Heartbuilder: BNIP-2 Influences Mechanosensing in Cardiomyoblast Differentiation
Researchers from the Low Lab at MBI discover a crucial role for the scaffold protein BNIP-2 in orchestrating focal adhesion dynamics during early heart development, offering new insights into heart regeneration strategies.
Bridging the (BP)GAP in metastasis
A collaborative study between researchers at MBI and scientists locally and overseas discovered how a scaffolding protein synchronizes, in space and time, two important regulatory proteins driving cell migration Learn more
Building a mechanobiological signaling scaffold for the heart
A collaboration between MBI scientists and clinical researchers revealed how a scaffolding protein integrates biochemical and mechanical signals to control cardiac muscle cell differentiation. Learn more
Spotlight on MBI Prof Low Boon Chuan: Unravelling the chemistry of life
A recent NUS News article features Assoc Prof Low, who leads a dynamic team at MBI focusing on how cells signal – the way they communicate with one another and within themselves.
A molecular brake
Recent study led by MBI Research Fellows Pan Meng and Ti Weng Chew and Principal Investigator Associate Professor Low Boon Chuan has revealed a role for BNIP-2 protein in scaffolding GEF-H1 and RhoA, following microtubule disassembly, and has described how this scaffolding is important for RhoA-dependent regulation of cell migration. Learn more
The mechanics of cancer growth
How cancer cells spread in response to physical cues from their surroundings
Low Boon Chuan
Principal Investigator
Research Areas
To identify novel signaling proteins and protein domains that control cell morphogenesis, motility, differentiation and cell growth and tissue/organ development during normal and disease states. Areas of interest include cell signaling, domain-discovery, protein-protein interaction, structural biology, developmental biology, computational biology and mechanobiology.
Research Interests
Molecular recognition forms the basis for all cellular events- from a simple bimolecular enzymatic reaction to the cascades of multimeric protein complex in cell signaling. Fundamental to the structure and function of a protein is its ‘domain’- a discrete, minimal modular entity that constitutes one of the basic physical and functional unit of the polypeptide. This protein domain can either serve as a protein docking/interaction site or an active enzymatic unit. With the emphasis on functional genomics, it is important to address what role does each of these domains play and how their potential functions can be regulated across molecular, cellular and tissue levels.
One of the several protein domains that our group first identified and characterised is a novel protein domain termed BCH domain which play important roles in regulating cell growth/death, differentiation, migration, and tissue/organ development. Based on the prototypical BNIP-2 and BPGAP1 proteins, we show that distinct BCH domains could act as key modulators for Rho and Ras small GTPases as well as their immediate regulators such as guanine nucleotide exchange factors and GTPase-activating proteins. Current effort is geared towards understanding how cells and tissues respond to the dynamic forces and geometry in the environment both under the influence of the BCH domain as a versatile regulatory scaffold domain. These effects will be examined under normal and pathological conditions such as in cancers, neurological disorders, skin and heart diseases.
Biography
Assoc Prof Low left Kuala Lumpur, Malaysia for Dunedin, New Zealand in early 90’s to pursue his dream as a scientist. Having spent wonderful undergraduate and postgraduate years at the Department of Biochemistry, University of Otago, he joined IMCB, Singapore and then NUS, trying to figure out what exactly make cells work. Amazing as it is, we are still far from understanding the intricacies underlying these processes. Trained as a biochemist, practising mainly as a cell biologist now, and with exciting arrays of multi-disciplinary tools, his team and collaborators aim to systematically unravel some of the uncharted paths, and are ready to expect the unexpected.
Selected Publications
- Wong DCP, Pan CQ, Er SY, Thivakar T, Rachel TZY, Seah SH, Chua PJ, Jiang T, Chew TW, Chaudhuri PK, Mukherjee S, Salim A, Aye TA, Koh CG, Lim CT, Tan PH, Bay BH, Ridley AJ, and Low BC. The Scaffold RhoGAP Protein ARHGAP8/ BPGAP1 Synchronizes Rac and Rho Signaling to Facilitate Cell Migration. Mol Biol Cell 2023;:mbcE21030099. [PMID: 36598812]
- Wong DCP, Xiao J, Chew TW, Pan M, Lee CJM, Ang JW, Yow I, Thivakar T, Ackers-Johnson M, Lee NJW, Foo RS, Kanchanawong P, and Low BC. BNIP-2 Activation of Cellular Contractility Inactivates YAP for H9c2 Cardiomyoblast Differentiation. Adv Sci (Weinh) 2022;:e2202834. [PMID: 35975420]
- Meng Pan, Ti Weng Chew, Darren Chen Pei Wong, Jingwei Xiao, Hui Ting Ong, Jasmine Fei Li Chin, Boon Chuan Low1. BNIP-2 retards breast cancer cell migration by coupling microtubule-mediated GEF-H1 and RhoA activation. Science Advances 2020.
- , , , , , , , , , , Hiltonol, a dsRNA Mimic, Promotes NK Cell Anticancer Cytotoxicity Through TAZ Cytoplasmic Sequestration. Adv. Therap. 2023, 2300016. https://doi.org/10.1002/adtp.202300016
Recent Publications
- Wu SK, Sun F, Ho CZ, Lou Y, Huang CB, Nai MH, Xiao J, Shagirov M, Chin JFL, Lim D, Verma S, Tan DSP, Marcq P, Yap AS, Lim CT, Hiraiwa T, Lin Y, and Low BC. Multiscale mechanisms driving tissue rupture by invading cells. Dev Cell 2026;. [PMID: 41785856]
- Ho CZ, Deng L, Picone R, Abderazzaq F, Flanagan N, Chua DZ, Low BC, and Wu SK. The Senescence-Associated Secretory Phenotype constitutes HIF-1α activation but is independent of micronuclei-induced cGas/Sting activation. Mol Biol Cell 2025;:mbcE24100445. [PMID: 41296514]
- Ho CZ, Lawther DB, Huang CB, Bharathkumar S, Beh SW, Poon EW, Lim PS, Lee KW, Felisha C, Tay SC, Yow I, Young JL, Wu SK, and Low BC. Protocol for AI-assisted quantitative analysis and setup of tumor spheroid invasion into tissue. STAR Protoc 2025; 6(4):104140. [PMID: 41075251]
- Sanz-Moreno V, and Low BC. Cell signaling across scales in health and disease. Curr Opin Cell Biol 2025; 97:102581. [PMID: 40946374]
- Xiao J, Ang JW, Zhong X, Wong DCP, T T, Yow I, Lee CJM, Foo RS, Kanchanawong P, and Low BC. Coordination of Focal Adhesion Nanoarchitecture and Dynamics in Mechanosensing for Cardiomyoblast Differentiation. ACS Appl Mater Interfaces 2025;. [PMID: 39778877]
- Shankar S, Liu Y, Tulsian NK, Low BC, Lin Q, and Sivaraman J. Insights into the regulation of CHIP E3 ligase-mediated ubiquitination of neuronal protein BNIP-H. PNAS Nexus 2024; 3(12):pgae536. [PMID: 39703232]
- Liu OX, Lin LB, Bunk S, Chew T, Wu SK, Motegi F, and Low BC. A ZO-2 scaffolding mechanism regulates the Hippo signalling pathway. FEBS J 2024;. [PMID: 39462647]
- Aureille J, Prabhu SS, Barnett SF, Farrugia AJ, Arnal I, Lafanechère L, Low BC, Kanchanawong P, Mogilner A, and Bershadsky AD. Focal adhesions are controlled by microtubules through local contractility regulation. EMBO J 2024;. [PMID: 38769437]
- Lee CJM, Autio MI, Zheng WH, Song Y, Wang SC, Wong DCP, Xiao J, Zhu Y, Yusoff P, Yei X, Chock WK, Low BC, Sudol M, and Foo RS. Genome-Wide CRISPR Screen Identifies an NF2-Adherens Junction Mechanistic Dependency for Cardiac Lineage. Circulation 2024;. [PMID: 38752370]
- Wu Y, Cheng J, Qi J, Hang C, Dong R, Low BC, Yu H, and Jiang X. Three-dimensional liquid metal-based neuro-interfaces for human hippocampal organoids. Nat Commun 2024; 15(1):4047. [PMID: 38744873]
Lab Members
Yee Zhuangli
Research Fellow, Low Group
Miguel SOON Jun Yi
PhD Student, Class of August 2025, Low Group
Soumya Bunk
PhD Student, Department of Biological Sciences
T Thivakar
PhD Student, Class of August 2023, Low Group
Srinivas Sheshagiri Prabhu
PhD student, Class of August 2022, Low Group, Bershadsky Group
Ang Jing Wen
Research Fellow, Class of August 2021, Low Group
Celestine Ze Qing Ho
Research Assistant, Low Group
Seah Sock Hong
Research Fellow, Class of January 2021, Low Group
Wu Yan
PhD Student, Class of August 2019, Low Group
Yow Ying Ming Ivan
Schimdt AI in Science Postdoctoral Fellow, Low Group
Recent Lab Alumni
Yong Yuen Chee Carmen
Research Assistant, Low Group
Jevon Tan
Research Assistant, Low Group
Khadijah Binte Zulkifli
Research Assistant, Low Group
Huang Yameng
Research Fellow, Low Group
Zuyang Aaron Fong
Research Assistant, Low Group
Tan Zi Yi Rachel
Graduate Student, Low Group
Shadame Yeo Shuet Ting
Research Apprentice, Low Group
Muhammad Faris Hazwan Bin Ramli
PhD Student, Class of August 2021, Young Group
Somsubhro Mukherjee
Alumni, Low Group
Wu Kaixiang Selwin
Senior Research Fellow, Low Group
Hamizah Cognart
Research Fellow, Low Group
Eugene Seow Qun Zhou
Alumni, Low Group